Science
Related: About this forumSolution of the Transport Equation in the Random Medium of High-Plutonium-Content HTR Lattice Cells.
Edit: Restored equation graphics.
The paper I'll discuss briefly in this post is this one: A New Collision Probability Approach for Solution of the Transport Equation in the Random Medium of High-Plutonium-Content HTR Lattice Cells (Indrajeet Singh,* S. B. Degweker,† and Anurag Gupta NUCLEAR SCIENCE AND ENGINEERING · VOLUME 189 · 101–119 · FEBRUARY 2018).
The problem of climate change has now advanced to a level such that future generations will be left with little choice but to attempt - it's uncertain whether this can actually be done, although it is possibly feasible - to reverse the addition of carbon dioxide to the atmosphere. It is notable that the sequestration of carbon dioxide - in the form of dangerous fossil fuels - took hundreds of millions of years, and thus any reversal will require intense engineering challenges, on a scale few of us can actually appreciate.
Let me give you a hint: Electric cars, solar cells, wind turbines and batteries won't cut it. These aren't even band-aids on the problem, never mind tourniquets; they're more like bringing styptic pencils to address the amputation of a leg.
The only feasible approach to address the engineering requirements will involve very high sustainable (24/7/365) temperatures which are only accessible using nuclear fuels.
The reactor described in this paper is not in a class that I personally favor, but it is capable of producing high temperatures. My problem with the reactor described herein is that it is designed to consume plutonium without generating new plutonium from depleted uranium. My feeling is that we must generate new plutonium at a rate much faster than it is currently being generated (largely in thermal spectrum reactors), which is roughly 100 MT per year. In order to completely eliminate all oil, gas, coal mining for energy, and, for that matter, uranium mining, and the vast quantities of copper, steel, aluminum mining required for the so called "renewable energy" industry - even at the trivial and useless levels at which it now exists - it is necessary to produce and fission (at 600 exajoules/year) 19 tons of plutonium per day. Therefore we cannot afford to consume plutonium without regenerating it.
Nevertheless, this paper addresses some interesting points about plutonium fuels that are worth contemplating:
From the introductory text, which discusses the type of reactor that dominates the current nuclear fleet, light water reactors, and, in a few places, notably India and Canada, the wonderfully neutron efficient heavy water reactors:
The paper assumes that all high temperature reactors are based on a type of fuel known as TRISO (for TRIStructural ISOtropic) fuel. In these fuels, actinides are suspended in a spherical carbon and silicon carbide matrix. It is not true that TRISO fueled reactors are the only option for producing high temperatures, but these types of reactors are generally the best known. These types of reactors are generally known as "pebble bed" reactors, early versions of which were more or less commercial failures with some exceptions, and in general they rely on the use of helium gas, which is not sustainable, world supplies of helium will have been depleted within a few generations: The gas released from party balloons boils off into space.
The paper describes the TRISO well enough as the introduction continues:
The authors note that the mean free path for uranium based fuels, including 233U obtained from thorium can be treated as a homogenous matrix, but this is not true for plutonium fuels:
...In this paper, we describe a new method developed for treating the thermal neutron transport in a random heterogeneous medium (which in the WIMS-D formalism contains the two large resonances of Pu that are not part of the resonance treatment method of this library). Themethod attempts to obtain exact expressions for the collision probabilities from one macroscopic mesh to another taking into account the random heterogeneous distribution of TRISO particles. Other than the statistical assumptions used to describe the medium, which fairly accurately describe the situations at hand, the method is exact.We present two independent derivations of the basic formulas. Both methods give identical expressions for the collision probabilities, which gives us confidence in the formulas so derived...
The paper, as one might expect, is mathematically heavy, some introductory flavoring:
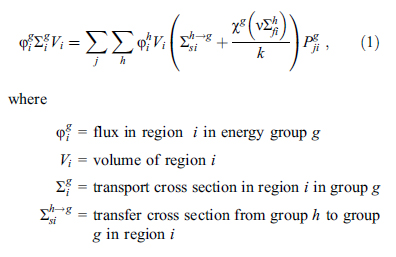

...ending up with this baby:

If this looks scary, don't worry be happy. The power of modern computers, which were not available at the dawn of the nuclear age, said dawn having produced some of the safest and cleanest energy devices ever commercialized (despite some very stupid press), specifically the pressurized water nuclear reactor, makes these sorts of calculations accessible.
This sort of thing is, of course, beyond the scope and understanding of dunderhead anti-nukes like, say, Amory Lovins, the Chief "Scientist" of the Rocky Mountain Institute, whose "Road Less Traveled" has become, regrettably, the "Road Most Traveled" with the result that as of this writing, we are seeing concentrations of the dangerous fossil fuel waste carbon dioxide in the atmosphere of over 413 ppm. This, as Mr. Lovins, says, is apparently "Winning the Oil End Game." Future generations will never forgive us for all this "winning," nor should they, since we took mindless assholes like Lovins seriously.
Anyway.
PMM is moderator to moderator probability. The reactor described herein is a moderated reactor which is designed to consume plutonium without regenerating it, something I find rather silly, since we need, again, more plutonium, not less of it. This reactor wastes valuable neutrons.
We do not need thermal (moderated) reactors to consume plutonium; we need fast (unmoderated) reactors to make more of it.
Speaking of plutonium, what I found interesting is the elemental composition of the fuel, specifically the isotopic distribution of the plutonium, shown in the following table:

This is "once through" plutonium, obtained from so called "nuclear waste" which is a silly term, since nothing that is extremely useful is actually "waste." At the very least what can be said for this type of putative reactor is that it at least utilizes plutonium as opposed to the very stupid idea of burying it as "waste."
Some other data relative to the Triso fuel composition and structure:

Some interesting data about the accumulation of americium and the interesting 242 isotope of curium, the latter representing a future source of helium, albeit, owing to the high energy density of nuclear fuels, nothing like the quantities we squander in this generation on balloons, at the expense of all future generations:

I hope you're enjoying the start of your weekend.
TexasTowelie
(112,531 posts)due to the weather. However, if it doesn't involve a triple integral or a differential equation then I'm disappointed. ![]()
NNadir
(33,579 posts)Equations 7 and 15 in the paper (not posted) are n-dimensional integrals.
TexasTowelie
(112,531 posts)I'll dn/dt, dp/dt, dx/dt, dy/dt and dz/dt all of that.
NNadir
(33,579 posts)
WheelWalker
(8,956 posts)NNadir
(33,579 posts)What I have learned of nuclear engineering is entirely out of an autodidactic means.
I would love to work at Hanford. I think about it all the time; one can learn some great stuff there about problem solving.
I did attend some wonderful lectures (in a non-nuclear area) by a very fine scientist at PNNL working in ion mobility mass spec - she has left to take a professorship at a University - and I had a few very interesting conversations with her on technical stuff related to the "exposiome" of the tissue concentrations of certain types of pollutants distributed widely across the planet in its atmosphere, and I suggested a nuclear application to her work, but that's as close as I got to Hanford.
My son, for the record, worked this summer at Oak Ridge, and got to tour the high flux reactor, which made me jealous, and also work with the neutron spallation source.
WheelWalker
(8,956 posts)He makes the same point as you about "once through" plutonium... that "Nothing that is extremely useful is actually 'waste'".
Reality is woven of opposing truths. You can recognize a deep truth by the feature that its opposite is also true. Blessings.
eppur_se_muova
(36,307 posts)Your remark about "wasting neutrons" led me to realize that this is what nuclear fuel really is -- neutrons. Neutrons are unstable, decaying to protons and electrons, with release of energy. Heavy elements contain more neutrons per proton than lighter elements, effectively captured in a "metastable" form by their association with protons, the heavier nuclei having more protons and thus capturing more than their share of neutrons. Nuclear reactors provide a mechanism by which these neutrons can be released from "storage" in heavy nuclei and, effectively, helped to decay (both directly and indirectly) to release their trapped energy.
The original source of these "excess" neutrons, of course, is the collapse of burned-out stars, which form heavy elements in supernova explosions. So nuclear reactors are fueled by collapsing stars, via the agency of heavy nuclei acting as neutron storage cells. I was just thinking of how disconcerted the early explorers of radioactivity were when they observed that a gram of radium salts produced enough heat to melt a gram of ice and convert it to steam within an hour, then repeat the process all over again, apparently indefinitely. No wonder they entertained doubts about whether the conservation of energy was absolute! Of course, knowing that mass and energy could be interconverted was the big key to understanding how so much energy could be stored so compactly. But the process of discovery was spread out over so many years I can't help but wonder if any of these researchers ever had that "eureka" moment when they could say "By Jove! I see it now! The enormous gravitational potential energy of stars is converted to the kinetic energy of infalling matter, which is converted to enormous thermal energy and compression of plasma/neutronium/whatever, some of which is used to create mass, and some of which is converted to the kinetic energy of an explosion, ejecting material against the pull of gravity (hello, planetary nebula!) and scattering neutron-rich "nutrients" to the cosmic soil from which the next generation of planets, complete with radium mines, forms! It all makes sense now!" ![]() Of course, maybe someone like Bethe did have that moment, but not many of the older researchers could have seen all these discoveries come together in their lifetime.
Of course, maybe someone like Bethe did have that moment, but not many of the older researchers could have seen all these discoveries come together in their lifetime.
Of course, I know you're already familiar with all that -- I'm just ruminating out loud, in the hope of cueing others to reach those same insights sooner in their lives than I did. That's what the progress of civilization is all about.
NNadir
(33,579 posts)External to a nucleus, neutrons are unstable to beta decay, and have a half-life on the order of 10 minutes.
In a nuclear reactor, the free neutrons have a very short mean free path, and depending on the various cross sections (which themselves depend on neutron energy) and interact with matter so frequently as to generally engage in some kind of nuclear reaction repeatedly in one of three ways in general, scattering, absorption, or fission. A 1 MeV neutron - fission neutrons emerge at energies with this order of magnitude, a traveling about 4% of the speed of light. They can hit a lot of nuclei, and depending on the mass of the nuclei, they can be significantly slowed, with lighter nucleic slowing them more effectively than heavy nuclei.
In the case of absorption, there are several types of materials with which they can interact, fission products (from earlier fissions), structural materials, moderators, or either fissile or fertile nuclei. (All fissionable nuclei can absorb neutrons without fissioning, and this important parameter, the fission to absorption ratio is very important in nuclear engineering. Fission cross sections depend on the energy of the neutrons. Nuclei normally not considered "fissionable," for example Pu-240, Pu-242, Pu-238, Am-241, Am-243, Cm-242, and Cm-244, can have appreciable fission cross sections when compared to absorption cross sections with "fast" neutrons, those with energies in the low single digit MeV energy range. Even U-238 has this property, although it cannot be made to reach a critical mass because of self-moderation. Still a fast reactor will directly fission some U-238, and the fission of U-238 in hydrogen bombs exploits this fact since fusion neutrons are extremely fast, 14 MeV.
When neutrons emerge from the core, they are said to "leak" and the leaking probability is an important factor in making a critical mass, which is why the shape of a mass has a profound effect on it's ability to reach criticality.
To me, if a neutron is absorbed in a fertile nucleus, thorium-232, uranium-238, or one of the "non-fissionable" actinide isotopes (at thermal energies) it is not "wasted." It transmutes an isotope into one that is fissionable in thermal neutron spectra, U-238 becomes U-239, which goes through two beta decays to give Pu-239, which is fuel.
If it leaks, or is absorbed in a moderator (water or carbon, generally) it's "wasted."
I believe we need sometimes to recharge certain radioactive elements to assure that they remain radioactive. Cesium, for example, contains three fission products, Cs-133 (stable), Cs-135, and Cs-137. The half-life of the latter is a little over 30 years. This means that cesium contained in used nuclear fuel generated in the late 1980's is only about half as radioactive as it was. From my perspective, it would be useful to put "old" cesium into neutron fluxes to generate the Cs-134 isotope (which is not a fission product, but represents induced radioactivity from capture in Cs-133) as well as Cs-136 and perhaps a little more Cs-137. The gamma radiation can then be used to destroy water borne and air borne recalcitrant halogenated pollutants, and, in fact, ground level ozone, a very damaging pollutant.
Neutrons that are absorbed into nuclei to induce radioactivity in nuclei that are not radioactive can be, under some circumstances, in my view, thus appropriately used neutrons. Finally, we may wish to absorb neutrons to make supplies of certain rare elements, technetium into ruthenium (although technetium is probably pretty valuable in its own right), tungsten into rhenium, iridium and osmium...etc...
Regrettably, because of Bateman equilibrium, we can never accumulate enough radioactive materials to address all of these diffuse pollutants, notably the carbon fluorides like PFOA and PFOS, and HFC's, but every little bit helps, I think. We can at least address the most seriously contaminated zones.
Neutrons absorbed into water, into steel, into zirconium, or other structural elements are "wasted."
A plutonium burning reactor which has no fertile nuclei and nothing useful to irradiate, wastes all of its neutrons, which is the case with the reactor described in the paper in the OP.
You are right though. All of the primary energy in the universe is nuclear energy, since all of the actinides were created in stellar explosions, as were most of the light elements except hydrogen, lithium, beryllium and boron.
I think it is clearly true that there would be no terrestrial life on Earth, were it not for uranium and thorium (and to a lesser extent radioactive potassium), since there would be no land without plate tectonics which is, ultimately, driven by the heat of nuclear decay.
The uranium and thorium we have are stored stellar energy, which we can recover, if we are smart.
I sometimes get insipid comments from people who say that the "only nuclear reactor I trust is the big fusion reactor in the sky."
In general these kinds of people are simply announcing that they are clueless.